Advances in Healthcare: Leadless Pacemakers
The latest advancement in healthcare technology for heart patients is the leadless pacemaker, which is aimed at eliminating the dangers of implanting pacemaker leads into the heart.
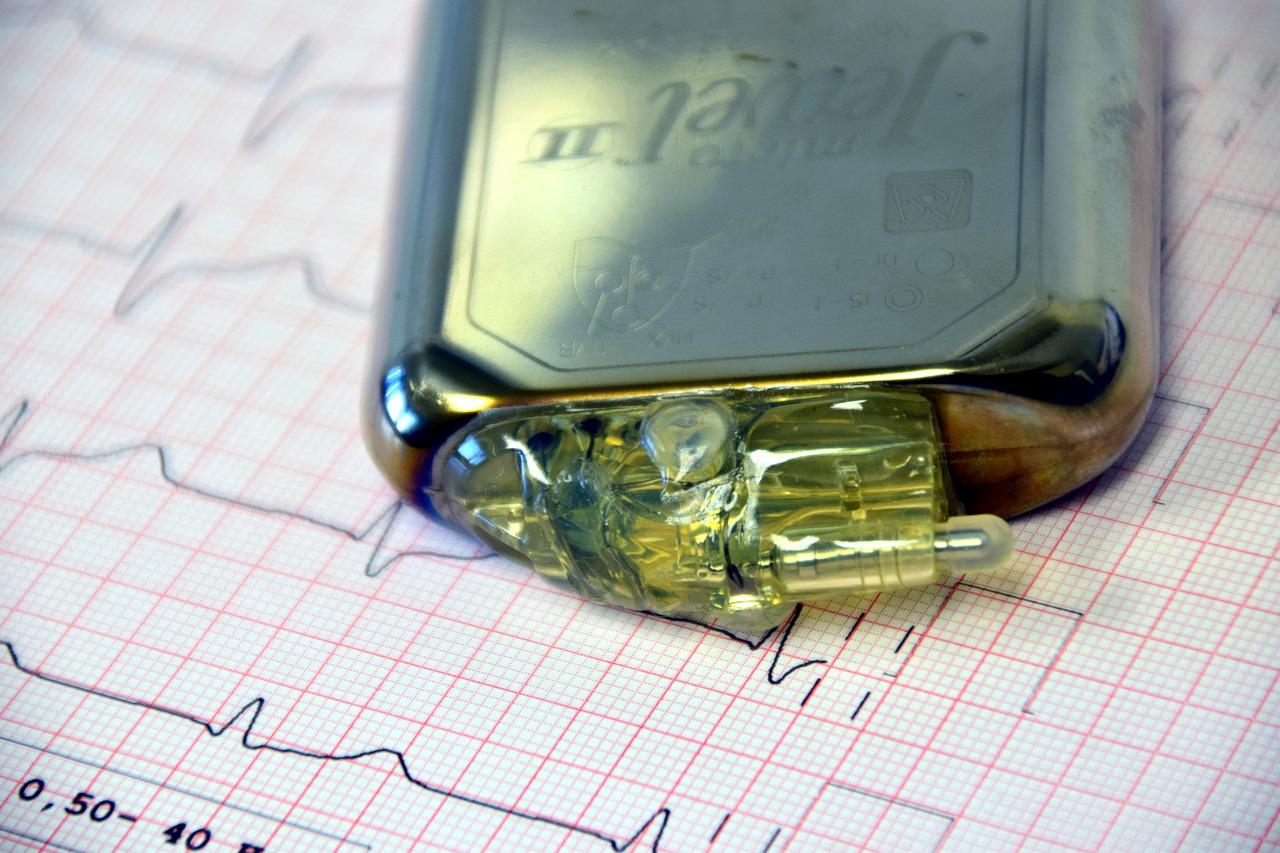
Traditional cardiac pacemakers are constructed using a Hypodermis Pulse Generator (in the chest wall) and transvenous pacing leads (in the myocardial tissue).
These leads are considered the weakest link of pacing systems as majority of early complications are related to lead placement, which include hematoma formation, cardiac perforation, and lead dislodgement.
Long-term complications include fracture due to mechanical stress, venous thrombosis, and infection of the pocket and bloodstream.
Completely implantable leadless pacemakers have recently become available for clinical use.
Leadless Right Ventricle Pacemakers
Proscribed for patients who require pacemakers in both the ventricles (chambers), fully implanted leadless pacemakers have been successfully implanted in patients who require pacemaker only in one ventricle.
It is also proscribed for heart patients who have existing cardiovascular implantable electronic devices (CIEDs), mechanical tricuspid valves, pulmonary hypertension, or IVC filters.
The present leadless pacemakers are considered fit for use even during MRI scans.
This article shall probe into a few leadless pacemakers that have been successful/unsuccessful.
Nanostim Leadless Cardiac Pacemaker
As per published journals, the Nanostim device has had a 97% success rate with more than 96% cases being free of any complication, and can be implanted safely with stable pacing parameters over the short term.
However, minute chances of RV perforation and cardiac compression during the implant exist.
In a safety-and-effectiveness trial, nearly 93% of subject patients were free from device-related serious adverse events.
Device related serious events were seen in only 6.7% of patients, including device dislodgment with percutaneous retrieval, cardiac perforation, and pacing threshold elevation requiring retrieval and device replacement.
Micra TPS
As per the results of a prospective multicenter trial, Micra TPC, a leadless pacemaker device, was successfully implanted in all 140 patients that were subjects in the trial, with early complications amounting to 18.6%, which include transient AV block, ventricular fibrillation and pericardial effusion without heart compression.
WiCS
A new WISE-CRT study was conducted with the insertion of WiCS leadless pacemaker device, which demonstrated improved LV function at six months in patients who were eligible for implantation.
However, since only about three quarters could be successfully implanted, the study was terminated prematurely. This device has been declared unsuccessful, but newer methods are being evolved to make it suitable for implantation.
Conclusion
Some challenges that these pacemakers must meet for future success include battery depletion and energy management.
Scientists are evolving newer methods at transforming Kinetic Energy from cardiac rhythms to power the pacemaker, which would tackle battery depletion or even render the use of battery obsolete.